Photo: Julia Baer. Fishing boat.
Fisheries
J. Baer, A. Smaal, K. van der Reijden & G. Nehls
Published 2017
1. Introduction
The Wadden Sea is an open ecosystem with close interconnections to adjacent North Sea waters. Consequently, neither the distribution of fish nor the main activities of fisheries and ecological consequences are confined to the inner Wadden Sea. The following chapters give an overview on the recent developments in the fisheries sector since the last Quality Status Report of 2009. The focus lies on the main target species: brown shrimp and blue mussel, which are dealt with in separate thematic reports . Additional fishing activities taking place in the Wadden Sea are summed up towards the end of this report. An overview of all activities, landings and licenses is found in Table 1. Today, the economically most important fisheries in the Wadden Sea are the North Sea brown shrimp (Crangon crangon) fishery and the shellfish fishery for blue mussels (Mytilus edulis) and, on a small scale, for cockles (Cerastoderma edule). Aquaculture activities focus on the cultivation of blue mussels on bottom culture plots and suspended seed mussel collectors in the Netherlands and Germany (Lower Saxony and Schleswig-Holstein).
Historial overviewAs fishing activities not only affect target species, but have wider impacts on marine ecological systems, the fishing development of the past century links up with current fishing practices in the Wadden Sea described in the following chapters. Looking at data on fishing effort in the North Sea during the past century gives an indication on the extent of change in the fish populations over a relatively short time frame. While fishing effort was still limited by shortage in fuel and coal supply prior to 1920, technological progress in the mixed and flatfish fisheries initiated a shift from beam trawling to the more effective otter board trawling. In the German Bight, fishing effort increased by a factor of 2.5 from 1924 to 1932. Annual landing figures in Germany almost tripled from before 1914 to the 1930s (Fock et al., 2013). After 1945, fishing capacity of the North Sea fleets increased in general and for beam trawlers in particular, resulting in high mortality rates, especially for haddock, cod and whiting (Pope & Macer, 1996). Fishing mortality of plaice averaged about 0.5 yr−1 during the 20th century, well above the rate of natural mortality of 0.1 yr−1 (Beverton, 1964). Throughout the period 1973-1999 cod, saithe, haddock and whiting contributed with more than 90 % of the landings, while total landings declined continuously as the stocks became depleted (Heath, 2005). In November 2000, ICES indicated that the cod stock in the North Sea was outside of safe biological limits and in serious risk of collapse (ICES, 2001). Many demersal fish species such as cod and plaice have complex life histories and various phases in their life cycles (juveniles, spawning females) can be particularly vulnerable to fishing. Plaice is exploited in the mixed flatfish fishery that is actually targeting sole. Due to the small mesh sized used, large numbers of undersized plaice are caught and discarded. This is problematic as the survival of plaice discards is very low (van Beek, 1989). This long-term exploitation of fish stocks has been found to affect growth, maturation and reproduction in a number of species. Generally, these high mortality rates cause earlier maturation and lower adult body size. Between the early 1900s and the mid-1980s, size at maturation for the north sea plaice decreased by up to 16 % (females) and 35 % (males); age at maturation decreased by up to 35 % (females) and 60 % (males) (Rijnsdorp, 1989). A recent study showed that the body weight of plaice at maturation decreased by 46 %, relative to the intensification of fishing around 1900 (Mollet et al., 2016). This change in life history traits and size structures, due to selective exploitation, can be expected to occur not just in the target fishery, but may apply to bycatch species as well (Mollet et al., 2016). |
2. Status and trends
2.1 Shrimp Fishery
Ecological background
Brown shrimp (Crangon crangon L. 1758) is a very common crustacean that inhabits the shallow waters along the East coast of the Atlantic Ocean from Norway to Morocco, the Baltic Sea, the Mediterranean and the Black Sea (Campas & Van der Veer 2008). The species is a key component in the Wadden Sea ecosystem and the coastal marine foodweb. Brown shrimps are omnivorous, acts as a predator of larvae and juvenile stages of several fish and benthic species, including infaunal organisms (bivalves, nematodes, oligochaetes), epifaunal organisms (amphipods, gastropods) and demersal organisms (shrimps and fishes) (e.g. Pihl & Rosenberg, 1984; Jensen & Jensen, 1985; Nilsson et al., 1993). It has been estimated that the shrimp population in the Wadden Sea area in the 1970s had reached the capacity to consume the entire macrobenthic production (Kuipers & Dapper, 1981). These high densities make it also a commercially very valuable species, which is solely used for human consumption. The brown shrimp ranks at the top three species caught from the North Sea with respect to landing value, which can exceed 100 Million Euro in some years (Temming & Hufnagl, 2014). Due to their large numbers, brown shrimps are also an important prey to a large variety of predators, mostly small fish (Redant, 1978; Tiews, 1978; Hislop et al., 1991). Two main fish species target brown shrimp of commercial size (> 50 mm) in the Wadden Sea, namely cod (Gadus morhua) and whiting (Merlangius merlangus) (Temming & Hufnagl, 2014).
Brown shrimps grow fast, but also experience high mortality. Reproduction occurs offshore at depths of 10-20 m (Tiews, 1954), egg bearing females can be observed all year round with the exception of September and October. Warm spring weather leads to a strong increase in juvenile abundance in shallow Wadden Sea waters in May and June. In August and September the new generation reaches commercial size which results in increased catches during this period. With migration of adult shrimps to deeper waters outside the Wadden Sea in December and January landings decrease. As shrimping starts fully again in March the previous years’ overwintered spawning stock is targeted and is closely related to previous years’ autumn landings (ICES, 2012).
Because of its life history, which is characterized by a short life span of maximally two years, the lack of an age structure in the population and a very long reproductive season, a formal stock assessment on age-based methods as applied for most other commercially fished species, is not possible for the brown shrimp (ICES, 2013). Understanding the pressures acting on the brown shrimp population is hampered by a number of uncertainties, such as gear efficiency, and a highly variable and poorly understood seasonal and spatial distribution (Siegel et al., 2005; Tulp et al., 2016). The lack of reliable stock size estimates is the fundamental difficulty when discussing sustainability and management strategies.
Landing statistics
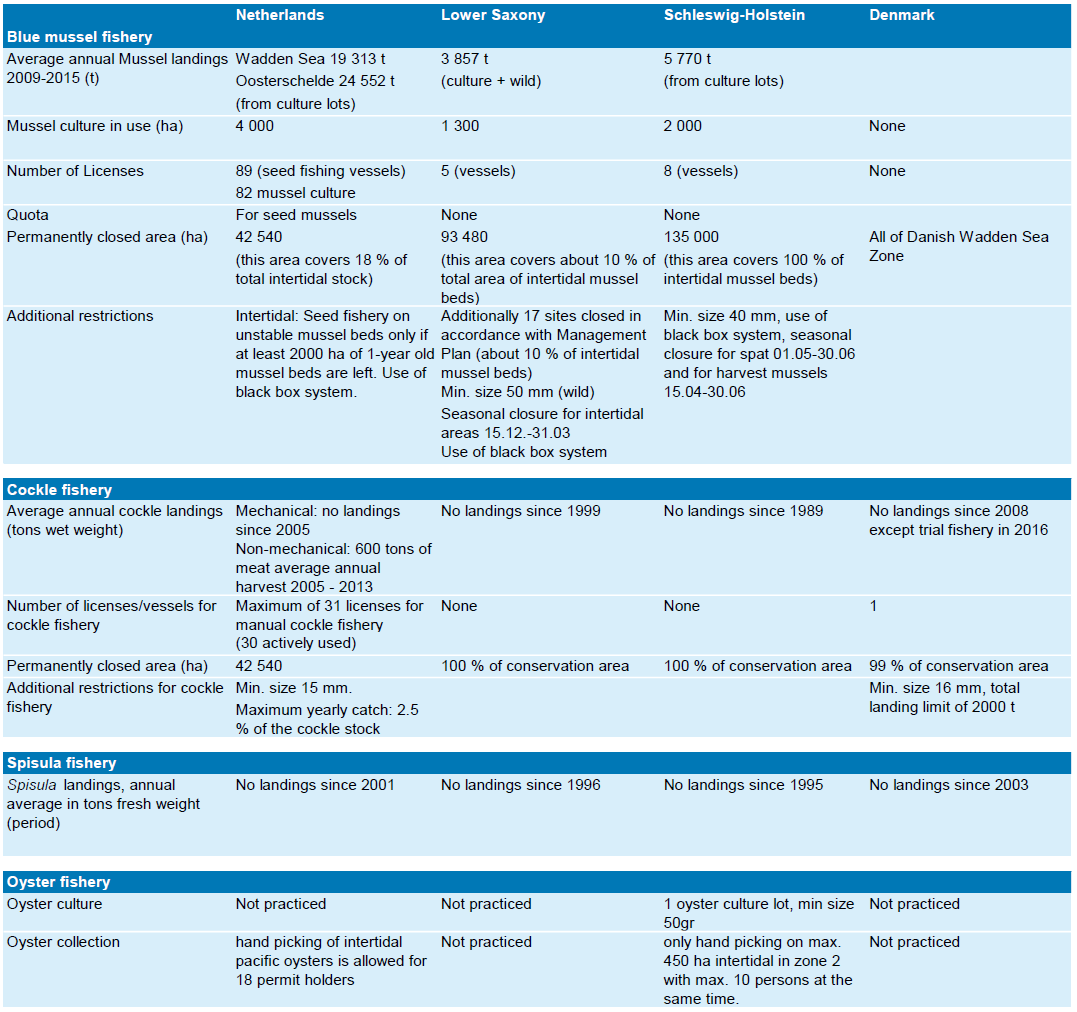
The total fleet targeting brown shrimps in the North Sea involves about 560 vessels, consisting of205 German, 200 Dutch, 27 Danish, 29 Belgian, 44 French and 54 UK shrimpers. The by far highest share is landed by the Dutch and German fisheries, which contribute 54 % and 33 % of the total North Sea landings, respectively. Over recent years the total amount of shrimps landed annually by German, Dutch and Danish fishermen constantly increased. While the share of German landings decreased continuously from 57 % in 1984 to the current 33 %, the share of Dutch landings increased from a minimum of 21 % in 1981 to a maximum of 54 % in 2014. The Danish share decreased since 2007 from 12 % to now 8 % (ICES, 2015). Total North Sea shrimp landings showed a seven-fold increase over the last fifty years and peaked in 2005 with 38,613 tons (Figure 1). The majority (>90 %) of the German cutters are smaller than 20 m, whereas in the Netherlands 60 % of the fleet is larger than 20 m. The engine power of German shrimpers is about 200 kW, with an increasing trend in engine power since 2002. About 60 % of the Dutch fleet is using engine power of more than 200 kW. The fishery concentrates on coastal areas of <30 m water depth (ICES, 2013). The highest ever reported brown shrimp landings, with 38,613 and 37,513 tons, were reached in 2005 and 2014, respectively (Figure 2).
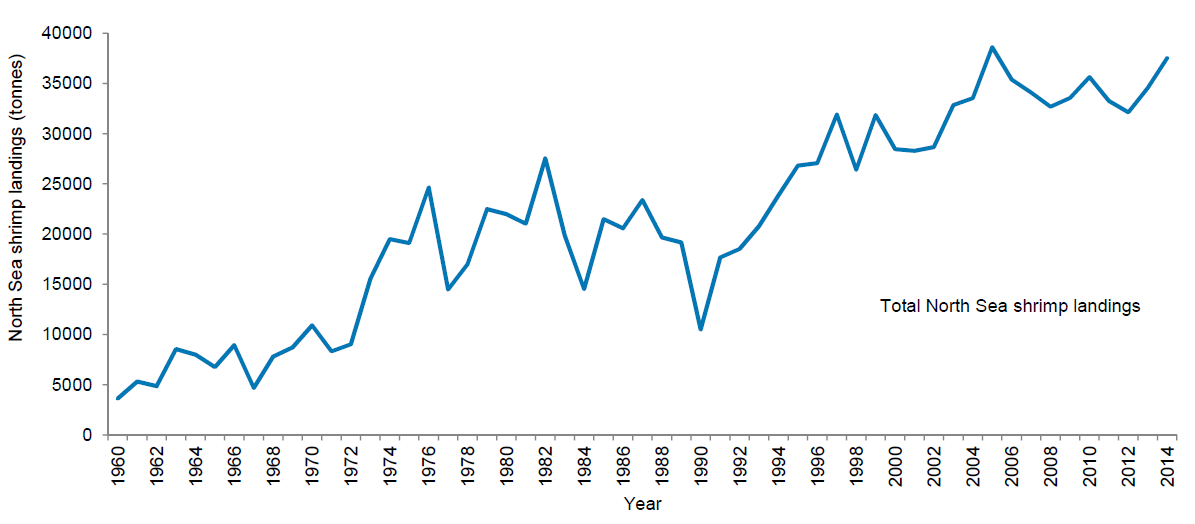 Figure 1. Development of total North Sea brown shrimp landings (t) 1960-2014 (ICES, 2015).
Figure 1. Development of total North Sea brown shrimp landings (t) 1960-2014 (ICES, 2015).
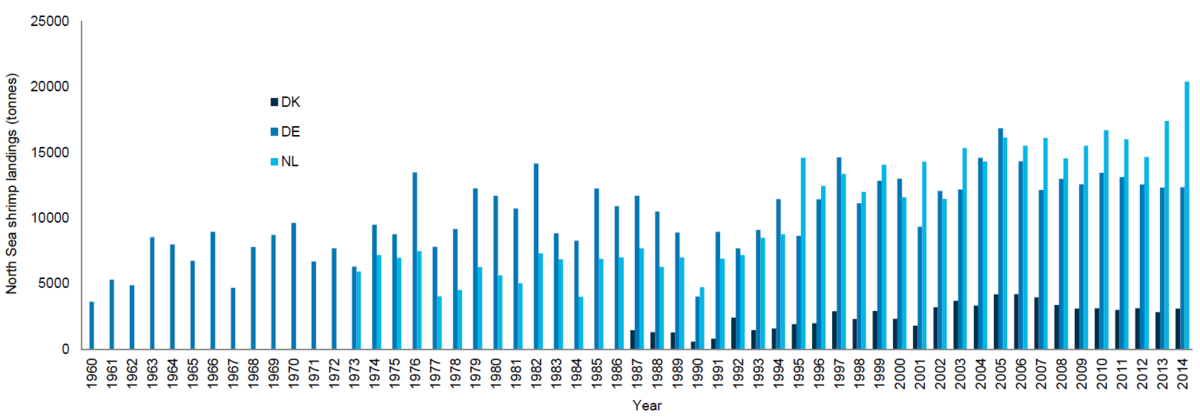 Figure 2. North Sea brown shrimp landings (t) for Germany, Netherlands, Denmark 1960-2014 (ICES, 2015).
Figure 2. North Sea brown shrimp landings (t) for Germany, Netherlands, Denmark 1960-2014 (ICES, 2015).
Data from satellite-based monitoring and reporting system for European fisheries (“Vessel Monitoring System”, VMS) allow the analysis of spatial and temporal distribution of the fishery. A recent study (WWF, 2016), in which a detailed analysis of VMS data between 2007 and 2013 was carried out, has shown that the focus of the German shrimp fishing fleet lies mainly on the exploitation of the coastal areas. Between 2007 and 2013 69.1 % of the fishing activities took place within the borders of the National Parks: Nationalpark Schleswig-Holsteinisches Wattenmeer 40.9 %, Nationalpark Hamburgisches Wattenmeer 0.3 % and Nationalpark Niedersächsisches Wattenmeer 27.9 %. Tidal basins, especially in the area of the Eider and Elbe estuaries, were also heavily targeted (25.8 %). This compares to only 5.6 % of shrimp fishing activity in offshore waters of the EEZ (seaward of the 12 nautical mile line). The analysis does not include foreign fishing vessels (Figure 3A).
VMS data from the Dutch fishing fleet in 2013 shows that fishing took place mainly in inshore waters off the Dutch coast and also within waters off the 3sm line of Schleswig-Holstein and Lower Saxony (Figure 3B).
Shrimping was introduced to the Danish Wadden Sea in the 1960s and comprised about 30 vessels in the late 1970s (Holm, 2004). Due to conservation concerns, the entire Danish Wadden Sea area was closed to fishing in 1977 and the regulation is still in force.
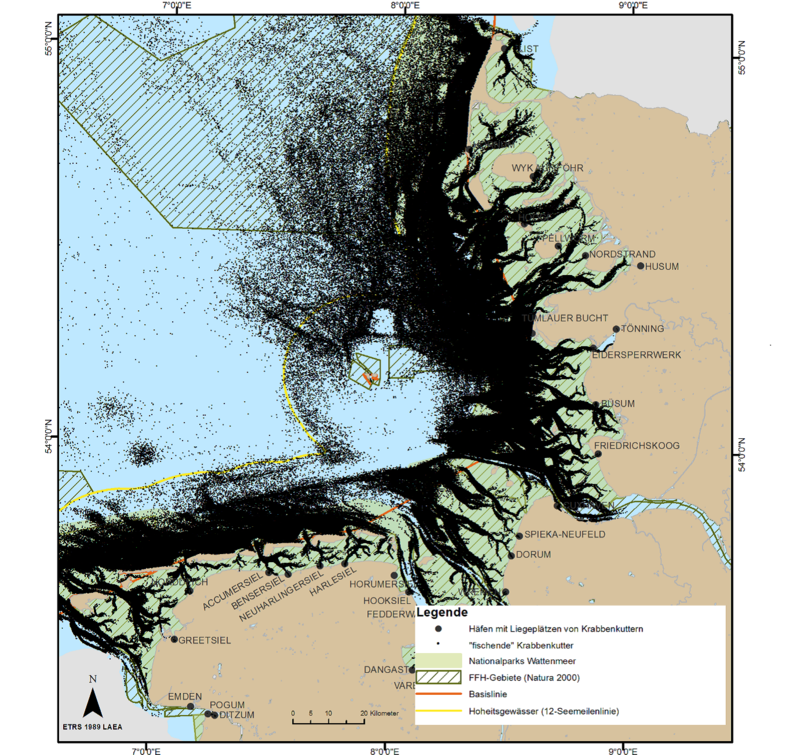
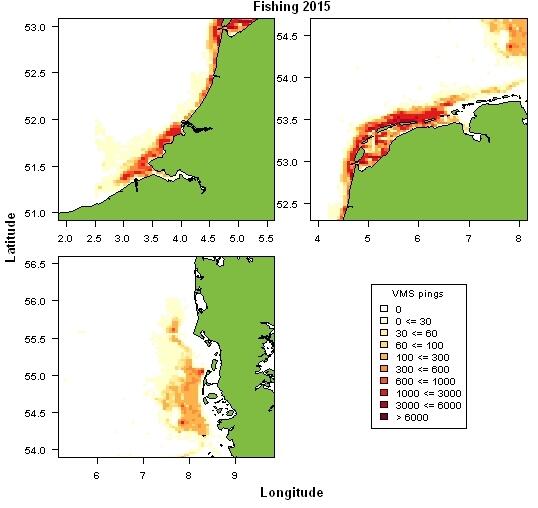 Figure 3. Left (A): German shrimping fleet distribution showing VMS data of actively fishing vessels 2007-2013 (Source: WWF, 2016); Right (B): Dutch shrimping fleet VMS (Vessel Monitoring System) data in 2013 (IMARES).
Figure 3. Left (A): German shrimping fleet distribution showing VMS data of actively fishing vessels 2007-2013 (Source: WWF, 2016); Right (B): Dutch shrimping fleet VMS (Vessel Monitoring System) data in 2013 (IMARES).
Management and sustainability
The North Sea brown shrimp fishery is currently neither regulated with quotas nor is the fishing effort limited by comprehensive government regulations, except by a restricted number of licenses, regional management initiatives (ICES, 2013; Tulp et al., 2016) and the VISWAD Covenant in the Netherlands (see below). The question whether brown shrimp management is needed, and how an efficient management could be, is a major debate in scientific circles, the shrimp fishing industry, on governmental level and NGOs. All parties are keen on improving economic and ecological sustainability of the shrimp fisheries and information on the status of the shrimp population is urgently needed. Shrimp beam trawling is carried out in vulnerable areas (coastal zones and estuaries), which are often nurseries for a wide range of marine species. A large extent of the shrimp fishing grounds also overlaps with Natura2000 sites, and therefore regional information on fishing activities and ecosystem effects is needed for management purposes.
Because brown shrimp is a key component in the marine food chain, taking into account the development of other fisheries and their possible impact on the shrimp population yields important information. The 1960s and 70s were characterized by large stocks of gadoid fish, the so-called ‘gadoid outburst’. In the subsequent decades this trend reversed due to combined effects of overfishing and climate change, causing a crash in cod and whiting populations (ICES, 2012; Myers et al., 1996). Temming & Hufnagl (2014) argue that increasing populations of harbour seal and grey seal exert further pressure on cod and whiting stocks, which are the most important predators of brown shrimp and their reduced numbers. Today, the shrimp fishery has become the main mortality source of adult brown shrimp, which is the result of the long-term increase of landings (Figure 1), with a risk of overfishing (Temming & Hufnagl 2014). With respect to setting management targets for brown shrimp, this presents a difficult situation, because the shrimp population is apparently enhanced from overfishing of fish stocks, mainly in the forefront of the Wadden Sea and the open North Sea, but also with contribution of bycatch from shrimpers. However, despite the human induced increase in the shrimp population, shrimp stocks show signs of fishing pressure as the increased fishing effort over the past years has not been correlated with increased landings and the fraction of large shrimp decreased over the past decades (ICES, 2015).
The main side-effects caused by trawl fishing are seafloor damage and bycatch. Mobile bottom fishing gears such as trawl fishing are used to catch species that live within, on or in close contact with the seabed such as scallops, shrimp and flatfish. As nets and dredges pass over the seabed, they damage the seafloor and remove a large biomass of the resident biota, often causing large-scale disruption in benthic habitats and communities. Collie et al. (2000) conducted a review of over thirty studies on the impact of trawl fishing and showed that the average immediate impact was the removal of 46 % of the individuals and a reduction in taxa of 55 % within disturbed plots. The magnitude of the response varied significantly with gear type, habitat and among taxa. So even when significant impacts can be demonstrated, the results usually pertain only to a particular gear on the given substrate and it is difficult to make general inferences on fleet level (see also O'Neill, F.G. & and A. Ivanović, 2016, for review).
Beam trawling typically targets different species at once due to its poor selectivity, resulting in a large amount of bycatch. The annual loss in landings arising from bycatch and discard by the shrimp fishery is estimated to have a market value of over 25 million euro (Revill et al., 1999). Currently, catches comprise about 30 % shrimps of commercial size, 30 % fish bycatch and 30 % undersized shrimps (ICES, 2015). The fraction of large shrimps (60 - 70 mm) decreased over time and varied between 13 and 18 % for 2013 and 2014, respectively. Plaice bycatch assessments show that the bycatch of the Dutch brown shrimp fleet alone accounts for 12-17 % mortality of the plaice spawning stock biomass (SSB).
Since 2009 an observer program in the Netherlands and Germany monitors the catch on shrimp trawlers and until 2012 a total of 167 hauls were sampled for catch and bycatch. It was found that in the Netherlands the total amount of shrimp landed was about equal to the amount of shrimp discarded (55 kg/hr landed and 56 kg/hr discarded). In Germany the amount of discarded shrimp exceeded the amount of landed shrimp by far (61 kg/hr landed and 105 kg/hr discarded). However, so far the sampling coverage of single fishing vessels is too low to be able to extrapolate these data to fleet level (Steenbergen et al., 2015).
The trilateral Wadden Sea Plan (2010) stipulates that in order to reduce bycatch and to reduce impact on the sea floor, trilateral policy principles for a sustainable shrimp fishery will be developed in close cooperation with the fisheries sector, a Framework for Sustainable Fisheries was decided at the 12th Trilateral Governmental Conference on the Protection of the Wadden Sea Tønder, 5 February 2014.
In the Netherlands, first steps were taken in spring 2013 towards closing parts of the Dutch Wadden Sea for shrimp fishery (and also mussel fishery, see sub-chapter “Management and sustainability”). Following this, the VISWAD Covenant (Covenant Shrimp Fisheries and Nature Ambition Rich Wadden Sea, consisting of representatives from government, shrimp fishery and NGOs) was formed in 2014 and aims to meet the demands as formulated for Natura 2000 (see www.rijkewaddenzee.nl). The ambition is to reduce the impact of the shrimp fishery on the ecological values in the Dutch Wadden Sea by 50 % before 2020. This will be achieved by a combination of measures such as reduction of fleet size, fishing permits, closing areas to shrimp fishery and changes of fishing techniques. Currently about 33,000 acres of the Dutch Wadden Sea are closed year-round to shrimp fishing (Figure 4). In August, an additional 16,000 acres are also closed to shrimp fishing, for the benefit of moulting shelducks (Tadorna tadorna). The next closing steps are expected in 2017.
In an effort to gain MSC certification for the North Sea brown shrimp fishery, a trilateral North Sea Brown Shrimp Management Plan has been adopted in 2015 by the fishery sector. The Management Plan is run by the Brown Shrimp Cooperative MSC Group, which consists of the Cooperative Fisheries Organization (Netherlands), the MSC-GbR for Germany and the Danish Fishermen – Producer Organization. Participation in the scheme is voluntary. Fishermen that decide to take part in the scheme must adhere to rules such as a defined number of hours at sea, no mesh size smaller than 20 mm and a limitation of undersized shrimp caught. A step-wise approach to the increase of the cod end mesh size from 22 mm, starting from May 2016, to 26 mm in May 2020 has been postulated as part of the management plan on sustainable yield. About 80 % of European shrimpers currently take part in the scheme.
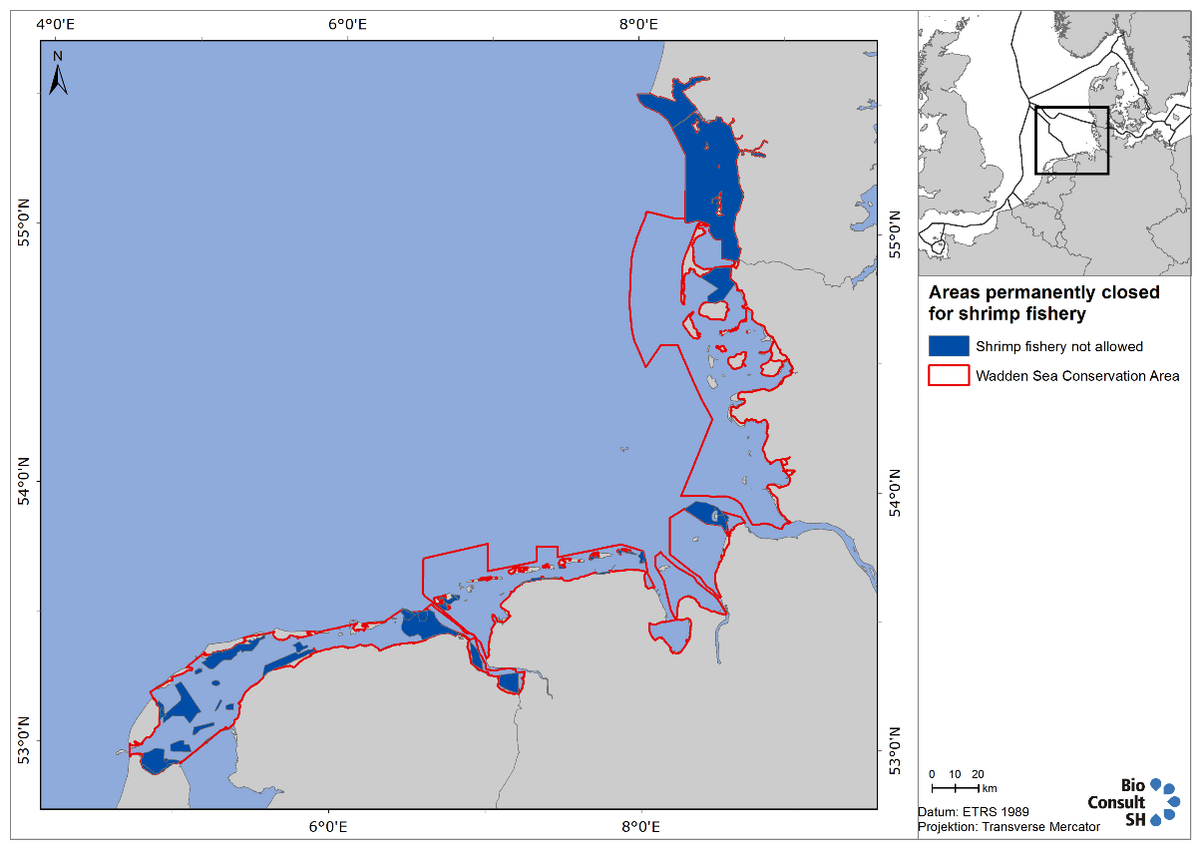 Figure 4. Map showing Wadden Sea areas closed year-round to shrimp fishery.
Figure 4. Map showing Wadden Sea areas closed year-round to shrimp fishery.
Alternative fishing methods
To help reduce discard of undersized shrimp and other bycatch species several trials with different mesh types and mesh sizes are ongoing. Currently standard cod end diamond mesh sizes of 20 mm are in use. Trials with diamond 26 mm and square 24 mm mesh sizes showed that undersized shrimp can be reduced by up to 80 % and fish bycatch by up to 20 % (Schultz et al., 2015). Bycatch of round fish species can even be reduced by rates of up to 60 % (Schultz et al., 2015). Population models showed that losses of marketable shrimp will be compensated by long-term population recovery, ultimately resulting in surplus catches.
A fishing method which is investigated as an alternative to traditional beam trawling is pulse fishing with electro trawls. In this fishery the mechanical stimulation by tickler chains or bobbins is replaced by electrical stimulation with electrodes. Low frequency pulse direct current (PDC) fields startle the shrimp, which causes it to leave the seafloor and enter the net. The removal of tickler chains or bobbins results in reduced bottom contact and drag, which in turn reduces fuel consumption and damage to the seafloor. With this, fishery bycatch can be reduced by up to 35 % with equal or increased shrimp catches (Verschueren et al., 2012) as the less intensive bottom contact reduces the amount of unwanted bycatch, especially flatfish, from being scooped from the bottom. However, in the current practice pulse fishing is carried out in combination with bobbin ropes (hard rubber rollers that keep the trawl in contact with the bottom), which makes it far more efficient in catching shrimp than the traditional beam trawls (Soetaert et al., 2015). An ecological advantage in terms of less bycatch and bottom contact per kg of fish caught is only reached if applied in combination with a limit in total allowable catch per year (ICES, 2015). Also, there are a number of unknown side effects of electrical fields on marine organisms, including their early life stages, which need to be investigated. For a review on pulse fishing and possible impacts see the 2014 WWF report and Soetaert et al. (2015). Since 2007 the European Commission granted Member States a derogation of 5 % of the fleet to use the pulse trawl on a restricted basis, but the EU’s Common Fisheries Policy allows projects “in support of avoiding unwanted catches in a fishery, which has led to an increase of vessels using the technique".
2.2 Blue mussel fishery and aquaculture
Ecological background and cultivation
Blue mussels are key components of the Wadden Sea ecosystem. They aggregate in mussel beds that occur both in the intertidal and in subtidal areas. Clumps of aggregated mussels originate from the ability of mussels to attach themselves to hard substrate by their byssus threads. This can be anything, from empty shells and small rocks to shells of other mussels. Other species may profit from the 3D structure created by the mussels as well, due to, in particular, shelter possibilities and wave dampening. The effects of mussels as ecoengineers in the ecosystem can reach up to 200 m outside the mussel beds (Donadi et al., 2013). Mussels also play an important role in the food web. As well-predated filter feeders, mussels are an important link between the primary producers and the higher trophic levels. Many birds, crabs, shrimps and fish (e.g. European flounder Platichthys flesus), are predating (a specific life stage of) mussels (Marsh, 1986, Nehls et al., 1997; Leonard et al., 1999; Borg et al., 2014; Heide et al., 2014).
Mussel cultivation in the Wadden Sea today is practised by means of mussel cultures by placing wild-caught mussel seed (spat) at specific sites, so-called on-bottom culture plots, where survival and growth is enhanced. Spat is originally collected by fishing on wild mussel seed beds, according to a fishing plan and on the basis of permits according to the relevant laws, or the seed is coming from Seed Mussel Collectors (SMCs, see sub-chapter “Alternative seed mussel collection technology”). To reduce the risk of import of non-native species, no imports of seed mussels have taken place from outside the Wadden Sea since 2008. However, in the Netherlands special licenses based on the Nature Protection law have been issued over the last few years to transfer mussel seed from the Oosterschelde to the Dutch Wadden Sea,. Yearly monitoring programmes are being executed in the Delta (SASI’s) to check problem-species. If those problem species have not been recorded, these so-called South-North transports could take place under certain conditions. Around mid-2015 these regular transports were ended due to the presence of the so-called ‘oesterboorder’ in the Oosterschelde. The Oesterboorder is a predatory sea snail (Urosalpinx cinerea) which is endemic to the Atlantic coast of North America and has been accidentially introduced with oyster spat in Northern Europe. Recently, a nature protection license has been granted to transport mussel seed, collected by smart farms in the Delta, to the culture lots in the Wadden Sea, under the condition that it has to be transported directly from the collectors, without any spreading time at the Delta-lots.
In Schleswig-Holstein, mussel spat is collected from July to October, with a seasonal closure period between 1 May to 30 June. In Lower SaxonyLower Saxony the seasonal closure time is restricted to intertidal areas, from 15 Dezember to 31 March. In the Netherlands, fishing for spat happens predominantly twice a year, in fall and spring. In fall, 0.5-year old spat is collected. In spring, wild mussel-seed beds are fished for 1-year old spat that survived the winter (Smaal et al., 2013). Culture stocks are maintained, for instance by the removal of the common starfish (Asterias rubens) (Barkhouse et al., 2007). Culture mussels are fished/ harvested several times and relayed on other plots, they are for instance brought to deeper located plots before the winter to protect them against storms and back in spring to the plots with the best growing conditions.
Depending on the strategy of the mussel farmer and the availability of mussel culture plots, the 1-year old spat (2-3 cm) is collected, transported and spread over new sites. Two years later, the mussels reach commercial sizes (4-5 cm). After harvesting, most mussels from Germany and the Netherlands are traded through the mussel auction in Yerseke (southwestern Netherlands) and spread out on so-called rewatering plots in the eastern part of the Oosterschelde (Netherlands). Here the mussels recover from transport and remain for a rinsing period prior to further processing, packaging and transport to the consumer.
Landing statistics
Mussel landings in Germany peaked over a period of 20 years between 1983 and 2003, after which landing values steadily decreased. In the Danish Wadden Sea area no mussel fishery has taken place since 2008 (Figure 5). A gradual decrease since the 90s can also be observed for mussel landings in the Netherlands (Figure 6).
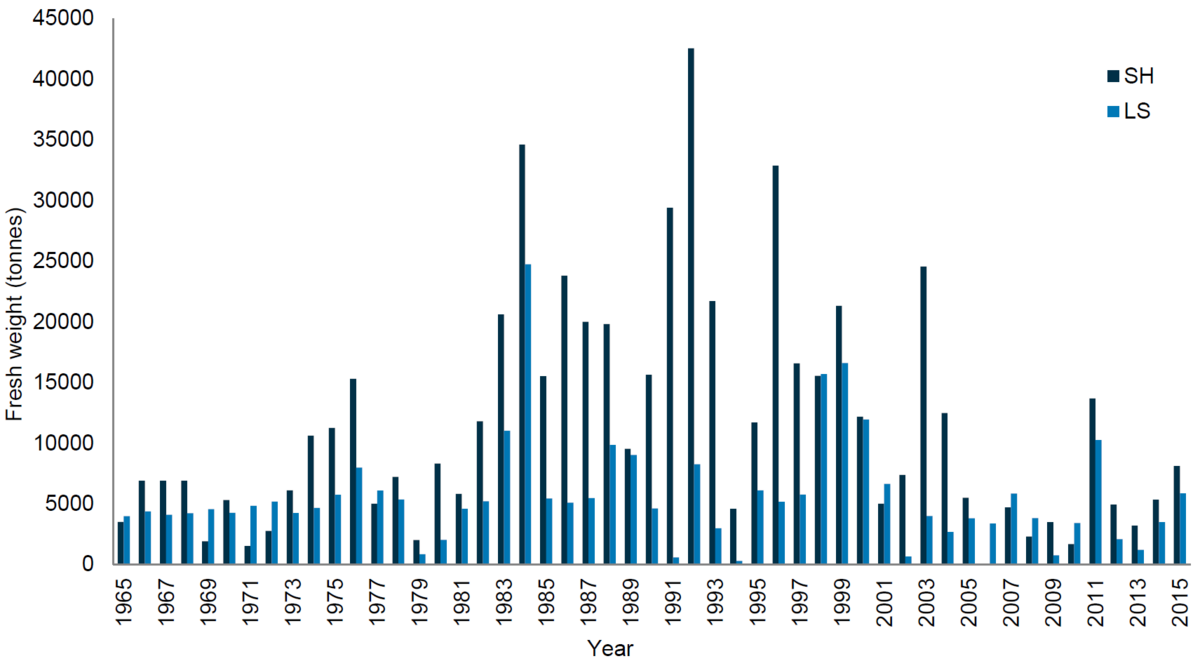 Figure 5. Development of mussel landings in Schleswig-Holstein (SH), Lower Saxony (NS) and Denmark (DK) 1965-2015.
Figure 5. Development of mussel landings in Schleswig-Holstein (SH), Lower Saxony (NS) and Denmark (DK) 1965-2015.
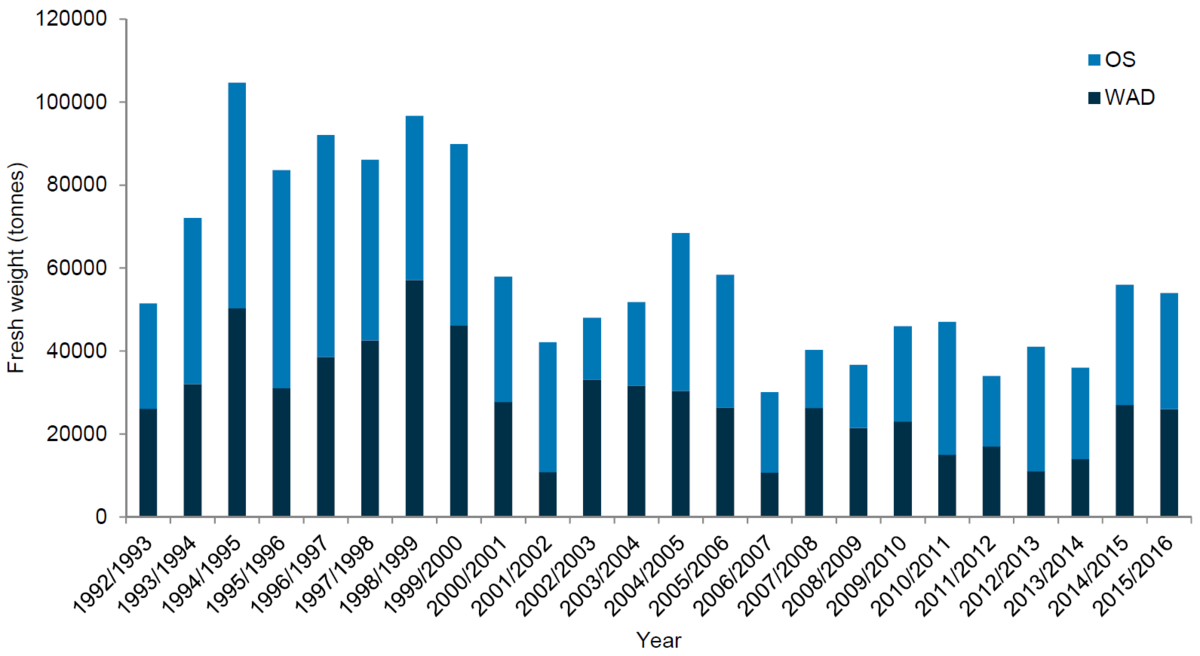 Figure 6. Netherlands: Mussel landing values in Wadden Sea (WAD) and Oosterschelde (OS, seed mussels derived from Wadden Sea) 1992-2016.
Figure 6. Netherlands: Mussel landing values in Wadden Sea (WAD) and Oosterschelde (OS, seed mussels derived from Wadden Sea) 1992-2016.
Management and sustainability
In the Netherlands, Denmark and Schleswig-Holstein, intertidal mussel fishery ceased in 1991, 1992 and 1995, respectively. Instead, seed mussels for stock culture lots have been obtained from subtidal areas only. In the Hamburg National Park mussel fishery has been prohibited since 1990. In Lower Saxony, fishery of seed mussels is allowed in the subtidal and defined parts of the intertidal. Therefore, direct impact of mussel fishery on intertidal mussel beds has been limited or absent during the last 20-25 years (de Vlas et al., 2005). Intertidal mussel bed areas have shown large annual variability, with successful recruitment as the predominant driver for re-establishment; closure of areas for fishery may help, but no direct relationship could be established (Ens et al., 2004). In Schleswig-Holstein and Lower Saxony mussel beds continued to deteriorate until 2005 despite reduced fishing impact; between 2005-2013 the areas of mussel beds remained constant but low. For a detailed analysis on the status of mussel surface area and biomass, see report “Beds of blue mussels and Pacific oysters”.
During the trilateral Tønder Conference on the Protection of the Wadden Sea (2014) a general framework and guiding principles on sustainable fisheries was agreed on. These guiding principles include trilaterally comparative impact assessments of all fisheries sectors in the Wadden Sea, facilitating a dialogue also between the industries and NGOs between the countries. The improvement of fishing gear, monitoring of fishing activities, stock assessments and pilot studies will also be trilaterally implemented to allow a Wadden Sea wide transition towards sustainable fisheries. The trilateral policies (Wadden Sea Plan 2010) state that mussel fishery will continue to be largely restricted to designated parts of the subtidal, with considerable areas left aside as feeding grounds for birds. Mussel culture lots will not be extended. Fishery practises are to be improved to further reduce impact.
Denmark
Mussel and cockle fishery has not been allowed in the Natura 2000 area in the Danish Wadden Sea since 2008 due to relative small stocks and the relative large amount of mussels reserved for mussel eating birds.
In 2013 a new Danish mussel policy has been implemented, which allows small-scale trial fisheries. Since September 2013 all permits (test or regular) for any fishery for mussels, oysters or cockles in Natura 2000 site require the installation of a black box system on each of the fishing vessels. The black box consists of a GPS-system, which logs the position of the boat every ten seconds, and a sensor on the winch that registers any activity e.g. starting or ending of fishing time. By monitoring the fishing activity and position, information on where the fisheries have taken place and also the extent of the areas affected are registered.
On the basis of the new policy, a permit for trial fishery of pacific oysters was initiated in 2015 to investigate if a cost-efficient and beneficial fishery could be established and at the same time fight the invasive pacific oyster. However, after only a few days of fishing the results showed that due to the reef-structure of the pacific oysters the fishery was not cost-efficient and was therefore stopped (pers. comm. SP. Jensen, The Danish Agrifish Agency).
Another trial fishery on cockles (Cerastoderma edulis) was carried out from 15 January to 29 February 2016. The cockle fishery was initiated on 3 February 2016 and restricted to three defined areas, where cockle fishery is allowed in the Danish Wadden Sea (BEK 867 of 21/06/2007). Total landing is limited to 2,000 t (wet weight) (pers. comm. SP. Jensen, The Danish Agrifish Agency). The analysis of the black box data has not been processed and therefore no further analysis of the cockle trial fishery has been reported yet.
Germany
Schleswig-Holstein
The current legislative basis for mussel fishery in Schleswig-Holstein is the mussel programme 2011 (Programm zur Bewirtschaftung der Muschelressourcen, § 40 Landesfischereigesetz). In February 2015 various stakeholders such as nature NGOs, representatives of the mussel fishing industry and the government came to an agreement on the future of mussel fisheries management in Schleswig-Holstein. While previously an area of 2000 ha was reserved for artificial mussel culturing, this area will be reduced to 1,700 ha. Of this area, 250 ha can be used as culturing areas for seed mussels (SMCs). All fishing activities will be recorded digitally on board and the information from the ‘black box’ will be made available to both the fishery authority and Nationalpark authority. Prior to this agreement, fishing for seed mussels was allowed within the subtidal part of zone 2 and two restricted areas in zone 1. According to the new agreement, mussel fishery will be restricted to four main tidal basins in the sublittoral of zone 2: Hörnumtief, Norderaue, Eidermündung and Piep (Figure 7). In the outer parts of zone 2 and in all of zone 1 activities connected with mussel fishery is prohibited. On the basis of this new agreement the fishermen apply for a 15 year valid permission and the required impact assessment is currently in preparation. The agreement provides the framework for the management of the fishery starting in early 2017. The MSC process by the Erzeugerorganisation Schleswig-Holsteinischer Muschelfischer e.V. has been successful and since 24 October 2016 the fishery is MSC certified, based on the 2015 framework agreement.
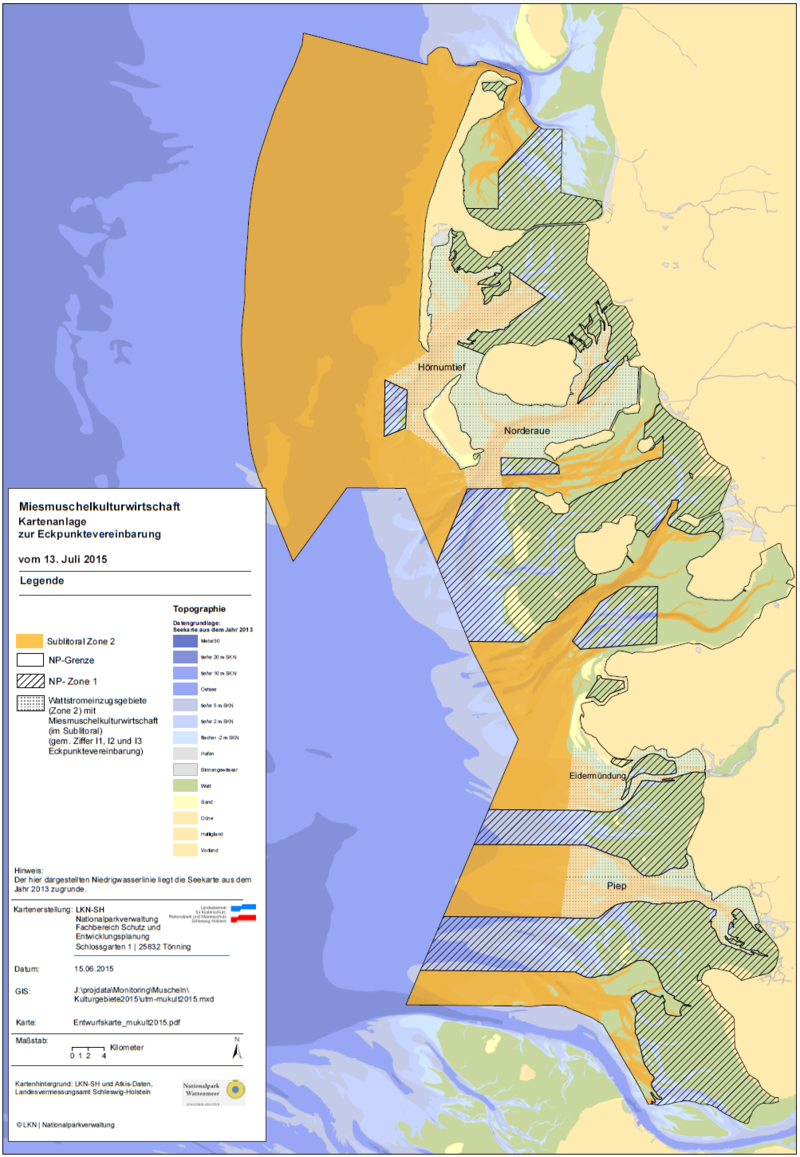
Figure 7. Areas open for blue mussel fishery (dotted areas) in Schleswig-Holstein according to the new framework agreement (Eckpunktevereinbarung zur Miesmuschelkulturwirtschaft, 2015).
Lower Saxony
The blue mussel fishery in Lower Saxony is managed via the ‘Blue Mussel Fishery Management Plan’ (BMMP), which is valid for 5 years and sets out conditions for the issuing of fishing permits and closure of stable intertidal mussel beds. The last management plan was established for the period 2009-2013 and a new management plan is in preparation. This new management plan aims to minimize the risk of introducing non-native species and stop the import of mussels from outside the Lower Saxon coastal area. All Niedersächsische Muschelfischer GbR member vessels have an additional black box installed, with which fishing activities can be monitored. The seed mussel fishery in the intertidal will continue to be allowed in areas where seed mussel settlement is temporary, as long as the condition on the minimum size of intertidal mussel beds is fulfilled: if the size of intertidal mussel beds (estimated by aerial survey) is at least 10 % below 1,000 ha and 10000 t biomass (reference values established in 1994), the Fisheries Office will not issue a permit for mussel fisheries on intertidal beds, until the 1994 reference values are reached again. Intertidal mussel bed biomass, size and locations are surveyed on an annual basis through aerial photography and ground-truthing. A sublittoral biotope survey is also planned.
There have been no major changes to the mussel management in Lower Saxony since 2009. The five licensed cutters harvest an area of 1,300 ha culture plots, which are mainly situated in the Ems and Jade Estuary (Figure 8). The whole area east of the river Weser and 34 % of the National Park area are closed for mussel fishery. Contrary to the other Wadden Sea areas, mussel seed fishery in the intertidal is allowed in some areas in Lower Saxony. Out of 102 stable intertidal beds, 29 have been closed for seed mussel fishery since 2004. In the subtidal all known concentrations of mussels can be fished under the present management plan. The use of ropes as substrate for seed mussels has been initiated in 2008. A total of five areas with Seed Mussel Collectors (SMCs) have been established, which are situated in close proximity to the Jade-Weser Port outside of the Wadden Sea Conservation area (Figure 8). As this technology requires a water depth of 5-6 m, most of the Wadden Sea area is not suitable. For more information on the technology of Seed Mussel Collectors (SMC) see sub-chapter “Alternative seed mussel collection technology”.
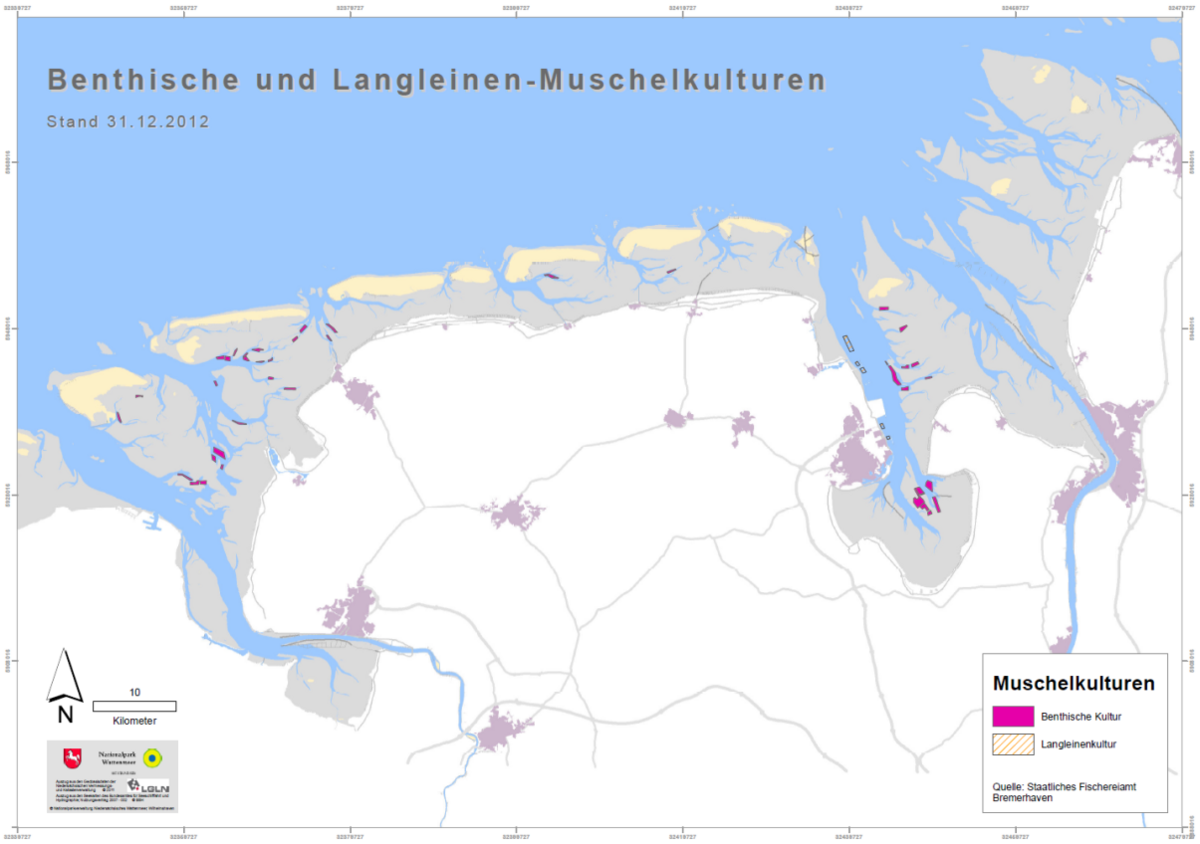 Figure 8. Position of the mussel culture plots (Benthische Kultur) and seed mussel cultures (Langleinenkultur) in Lower Saxony (Map: Nationalparkverwaltung Niedersächsisches Wattenmeer, Data source: Staatliches Fischereiamt Bremerhaven, 2012).
Figure 8. Position of the mussel culture plots (Benthische Kultur) and seed mussel cultures (Langleinenkultur) in Lower Saxony (Map: Nationalparkverwaltung Niedersächsisches Wattenmeer, Data source: Staatliches Fischereiamt Bremerhaven, 2012).
In 2013 the five licensed mussel fishery vessels gained MSC certification. Connected with the certification are a number of measures, which are also in parts subject to the new management plan, i.e. the mussel banks must show at least a size of 1.000 ha and a biomass of 10,000 t live weight, or the fishing license will be suspended and the intertidal areas are closed for fishing between mid-December and the end of March. Additionally, all fishing activities have to be documented digitally in a ‘black box’ and no import of seed mussels from outside the Wadden Sea is accepted. As part of the MSC certification, the fishermen have committed to carry out a research project to find out, whether further stable mussel beds can develop in the Wadden Sea of Lower Saxony.
Netherlands
The covenant, agreed in October 2008 between the mussel producer organisation, the Wadden Sea environmental NGOs and the responsible Dutch Ministry, has set the basis for the actual policy. It was agreed that over the period 2009-2021 bottom seed fisheries will be stepwise reduced, as a function of the development of alternative seed resources such as Seed Mussel Collectors (SMCs) (see also next sub-chapter). It was also agreed that subtidal mussel seed bed areas that will be closed for seed fishery (Figure 9), will also be closed for other types of fishery, and that mussel culture plots will be optimized in order to improve culture yield as a basis for return of the investments in SMCs.
The mid-term evaluation in 2014 has shown the following:
- 9,200 ha mussel bed area were closed for mussel fishery (cumulative in Waddensea, Oosterschelde and Voordelta)
- Seed production through SMCs was successful: the yield increased steadily from 1 mln kg in 2006 to 15 mln kg in 2014. The yield from the Dutch Wadden Sea in 2015 was around 16 mln kg (van Stralen, 2016).
- The cost of SMC seed for the mussel farmer was estimated as 0.8 €/kg, in comparison to bottom seed for a cost of 0.1 €/kg (LEI, 2014)
- More effort is needed for maintaining or improving culture plot quality, as geomorphological changes over time in the Wadden Sea have made culture areas less suitable. Given the price of SMC seed, this is considered as an urgent issue
- For efficient use of SMC seed, transport of seed from the Oosterschelde to the Wadden Sea should be made legal, as long as the risk of introduction of invasive species can be avoided
- The outcomes of the seed fishery impact study have shown that subtidal wild mussel bed survival is limited, also without fishery. Seed fishery in autumn had no significant impact on the seed stock and on biodiversity as mussel seed in the unfished control plots disappeared at a similar rate as on the fished plots only a few months later. The impact of fishery in the following spring sustained for two years after fishery for the seed stock and for one year for biodiversity. The study also showed that mussel culture plots had a biodiversity similar to wild beds (Smaal et al., 2013).
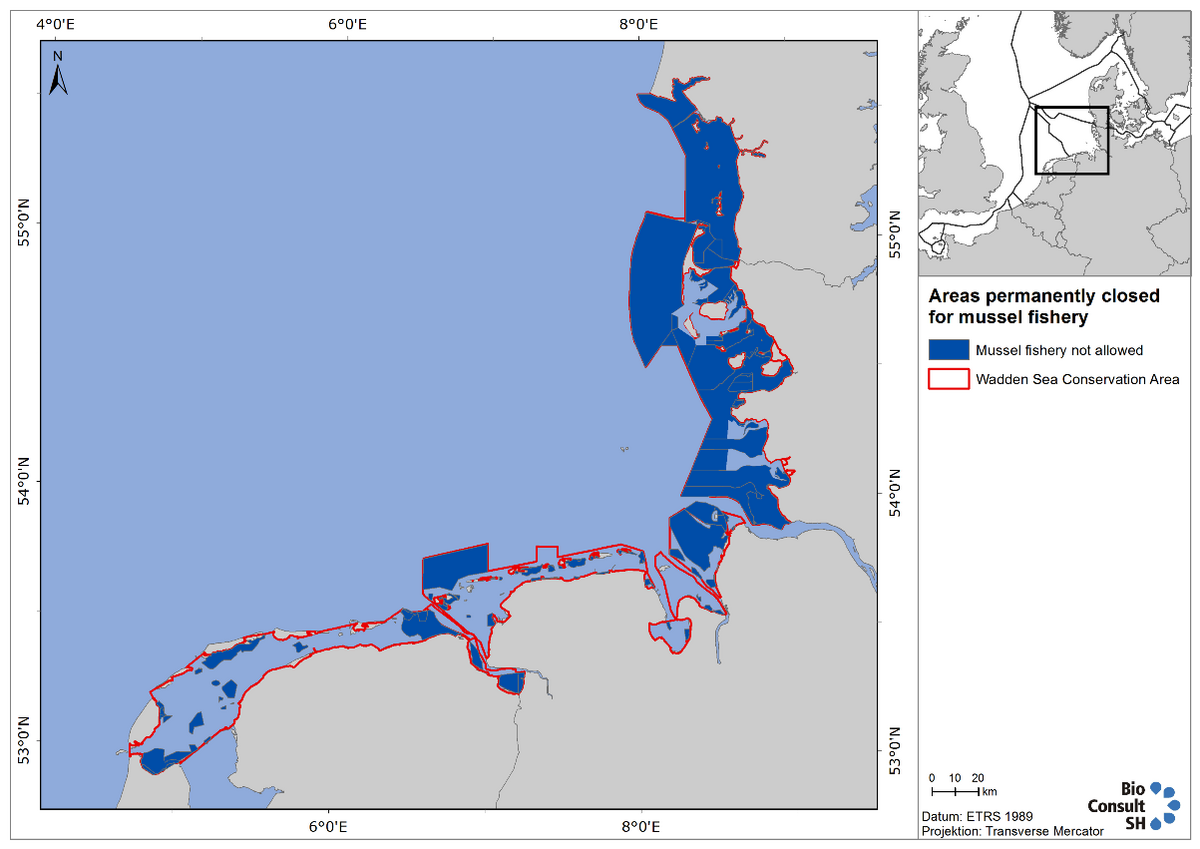 Figure 9. Wadden Sea areas currently closed to mussel fishery. This map includes future closed areas in Schleswig-Holstein when the updated management plan comes into effect.
Figure 9. Wadden Sea areas currently closed to mussel fishery. This map includes future closed areas in Schleswig-Holstein when the updated management plan comes into effect.
Alternative seed mussel collection technology
The occurrence and density of mussel beds is highly variable. Natural conditions that can cause mussel beds to disappear are mainly insufficient spatfall, ice covers and storms (Nehls & Ruth, 2004). Despite the fact that a large number of mussel larvae can be found in the water column in early spring, mass settlement of young mussels in the Wadden Sea is a rare and fluctuating event. Spatfall is a crucial and yet poorly understood driver in mussel population dynamics, which can be highly variable on small spatial scales (de Vlas et al., 2005). Climatic changes also appear to affect mussel recruitment. Cold winters can be of advantage for young mussels since predator populations (i.e. shore crabs Carcinus maenas, brown shrimp Crangon crangon and sea stars Asterias rubens), need longer to recover in the spring time, resulting in a head-start for the settlement of mussel larvae (Beukema & Decker, 2005). However, cold winters have become a rare event in the past 20 years and recent mussel recruitment has been low compared to the 1970s and 80s (Walter & Leeuw, 2007). Another factor influencing mussel bed development is the establishment of the non-native pacific oyster Crassostrea gigas, which competes with blue mussels for food and space and is characterized by high growth rates and successful recruitment (Damm & Neudecker, 2006; Büttger et al., 2013). Mussel beds with a high coverage of pacific oysters often become unsuitable for commercial mussel harvesting.
Mussel seeds are usually caught from the wild spat stock with the use of a mussel dredge. The dredge consists of a metallic net that is supported by a steel bar frame. The net is usually 1.9 m wide and a steel bar of 4 cm is dredged along the sea bottom and is emptied into the boat hold once filled to capacity. In Lower Saxony both dredge and trawl nets are used, depending on the characteristics of the sea floor and the size of the mussels. Because of these often low and spatially fluctuating spatfall events and to reduce the impact on the seafloor, the focus of the mussel fishing industry has turned increasingly towards artificial seed collection technologies (e.g. SMC). So called long lines or rope/net seed collectors have been on trial and commercial production outside Europe for a number of years. In the Netherlands and Germany the SMCs have been applied since around 2000. In these ‘Smart Farms’ ropes or nets are suspended from buoyant PE-pipes as growth substrate for seed mussels (Figure 10).
After the initial experimental phase, the use of this technique has been intensified. In Schleswig-Holstein (ca. 61 ha in Hörnumtief) and in Lower Saxony (ca. 173 ha over 5 areas, see Figure 8) are currently used for seed mussel collectors. However, there are some restrictions to the application. Sufficient water depth and close proximity to harbours are necessary, as all gear has to be brought to shore during winter and is deployed again in spring. In the Netherlands the SMC technology is widely used, with comparatively large areas, which have been allocated by the government. The total area in use in 2015 was 339 ha, of which 239 ha are located in the Dutch Wadden Sea, 83 ha in the Oosterschelde and 17 ha in the Voordelta (Figure 11A), with continuously increasing yields from different areas (Figure 11B). An extensive SMC impact study has addressed potential effects on the carrying capacity for protected species, such as birds, that prey upon bivalves and that may be affected by SMCs. Also the effects of disturbance through labour on the SMCs were addressed, including drilling of anchor pipes, as well as effects on the underlying sediment. Although no negative impacts were detected on short term, a monitoring programme has been installed to further explore long term effects (Kamermans et al., 2013).
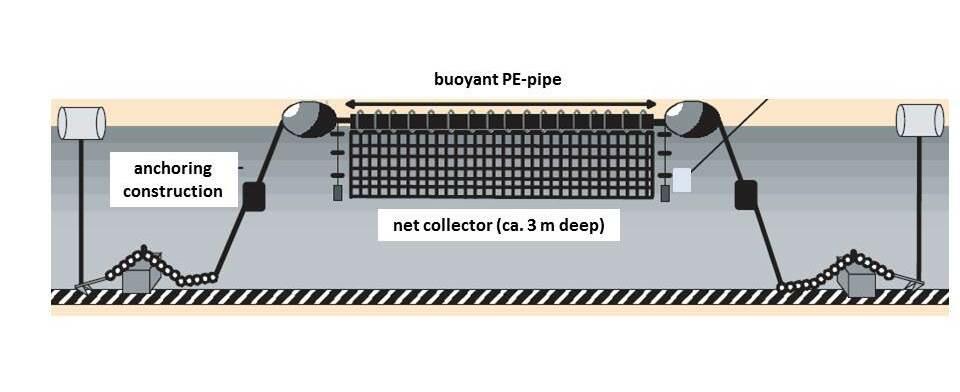 Figure 10. Common design of mussel seed collectors (after Walter & de Leeuw, 2005).
Figure 10. Common design of mussel seed collectors (after Walter & de Leeuw, 2005).
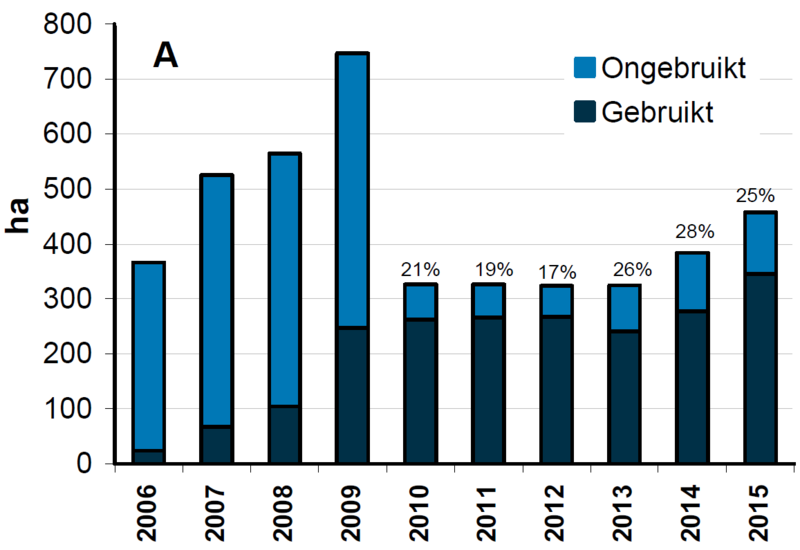
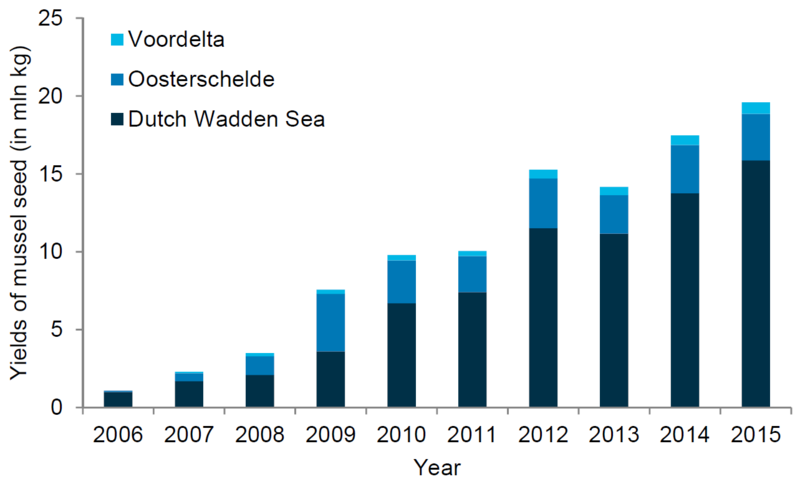 Figure 11. Left (A): Area reserved and in use for SMC’s in the Netherlands, over time (van Stralen, 2016); Right (B): Yield of mussel seed in million kg fresh weight from SMC’s in Dutch Wadden Sea, Oosterschelde and Voordelta over time (van Stralen, 2016).
Figure 11. Left (A): Area reserved and in use for SMC’s in the Netherlands, over time (van Stralen, 2016); Right (B): Yield of mussel seed in million kg fresh weight from SMC’s in Dutch Wadden Sea, Oosterschelde and Voordelta over time (van Stralen, 2016).
2.3 Other shellfish fishery
Denmark
All forms of other shellfish fishery are prohibited within the Danish Wadden Sea Zone.
Germany
In Schleswig-Holstein, the only shellfish harvested other than the blue mussel, is the Pacific oyster (Crassostrea gigas). While there is one oyster aquaculture plot of 30 ha, collection by hand is allowed on max 450 ha in zone 2 of the National Park by max. ten people at the same time. The thick trough shell (Spisula solida), has not been harvested since 1996 as their numbers have been too low. The fishing rights were not extended after 31 December 2016.
There is no shellfish exploitation Lower Saxony other than the blue mussel fishery in Lower Saxony. In the Hamburg National Park all shellfish fishing is prohibited.
Netherlands
In the Dutch Wadden Sea, a variety of fisheries other than blue mussel and shrimp fisheries are known. Most common are the manual cockle (Cerastoderma edule) fishery and fisheries using passive gears, like gillnets, fykes and pots. For cockle fisheries 31 permits have been issues for handraking of cockles in designated areas (Table 1). Annual harvest should not exceed 5 % of the overall stock (PMR, 2011) which is controlled during annual surveys. Annual licenses are issued on basis of the Nature Protection law i. The stipulated quota assigned to permit holders amounts to 2.5 % of the total amount of cockles available at densities above 50/m2 on 1 September. Over the last years, 18 permits for handraking of oysters (Crassostrea gigas) have been issued (by the province of Fryslân), based on the corresponding Nature Protection law for handraking of oysters.
These fisheries are described in more detail in a (Dutch) report (Overzee et al., 2008) and updated and summarized in a (Dutch) fact-sheet (Tulp et al., 2012).
Table 1. Facts on current shellfish fishing (source QSR 2009, updated).
3. Summary
In contrast to the outer North Sea, where mainly fish is targeted by large vessel or small scale fishery, the fisheries in and adjacent to the Wadden Sea concentrate on shrimps and mussels. Blue mussels are the most intensely exploited bivalve shellfish in the Dutch and German Wadden Sea, also by using cultures or mussel seed transfers. This fishery is strongly regulated and vast areas are closed for fishing activities, with different extent over the three countries. Denmark, for instance, has banned shrimp and mussel fishery entirely. Alternative techniques with potentially less impact to the ecosystem are under development, as well as corresponding national management and protection regimes.
About the authorsJ. Baer1, A. Smaal2, K. van der Reijden2 & G. Nehls1 1 BioConsult SH GmbH & Co.KG, Schobüller Str. 36, 25813 Husum, DE 2 Wageningen University, IMARES, Korringaweg 5, 4401 NT Yerseke, NL |
References
Barkhouse C.L., M. Niles, & Davidson L.-A. (2007) A literature review of sea star control methods for bottom and off bottom shellfish cultures. Can. Ind. Rep. Fish. Aquat. Sci. 279: vii + 38 p.
Beukema J.J. & Dekker R. (2005) Decline of recruitment success in cockles and other bivalves in the Wadden Sea: Possible role of climate change, predation on postlarvae and fisheries. Marine Ecology Progress Series 289: 149-167.
Beverton R. J. H. (1964) Differential catchability of male and female plaice in the North Sea and its effect on estimates of stock abundance. Rapports et Procès-Verbaux des Réunions du Conseil Permanent International pour l’Exploration de laMer, 155: 103–112.
Borg J.P.G., M. Westerbom & Lehtonen H.(2014) Sex-specific distribution and diet of Platichthys flesus at the end of spawning in the northern Baltic Sea. Journal of Fish Biology 84(4): 937-951.
Büttger H., Nehls G. & Stoddard P. (2013) The history of intertidal blue mussel beds in the North Frisian Wadden Sea in the 20th century: Can we define reference conditions for conservation targets by analysing aerial photographs? Journal of Sea Research 87: 91–102.
Campos J. & Van der Veer H.W. (2008) Autecology of Crangon crangon (L.) with an emphasis on latitudinal trends. Oceanography Marine Biology Annual Review 46: 65-104.
Collie J. S., Hall S. J., Kaiser M. J. & Poiner I. R. (2000) A quantitative analysis of fishing impacts on shelf-sea benthos. Journal of Animal Ecology, 69: 785–798.
Damm U. & Neudecker T. (2006) Zweikampf im Wattenmeer. Wird die heimische Miesmuschel verdrängt? Informationen aus der Fischereiforschung 53: 46-48.
de Vlas J., Brinkman B., Buschbaum C., Dankers N., Herlyn M., Kristensen P. S., Millat G., Nehls G., Ruth M., Steenbergen J. & Wehrman A. (2005) Intertidal Blue Mussel Beds. Wadden Sea Ecosystems 19: 190-200.
Donadi S., t. van der Heide E.M. van der Zee, Eklöf J.S., v. de Koppel J., Weerman E.J., Piersma T., Olff H. & Eriksson B.K. (2013) Cross-habitat interactions among bivalve species control community structure on intertidal flats. Ecology 94: 489–498.
Ens B.J., Smaal A.C. & de Vlas J. (2004) The effects of shellfish fisheries on the ecosystems of the Dutch Wadden sea and the Oosterschelde. Alterra report 1011, Wageningen.
Fock H. O., Kloppmann M.H.F., Probst W.N. (2013) An early footprint of fisheries: Changes for a demersal fish assemblage in the German Bight from 1902–1932 to 1991–2009. Journal of Sea Research 85:325–335.
Hislop J.R.G., Robb A.P., Bell M.A. & Armstrong D.W. (1991) The diet and food consumption of whiting (Merlangius merlangus) in the North Sea. ICES Journal of Marine Science 48: 139-156.
Holm P. (2004) Human impacts on fisheries resources and abundance in the Danish Wadden Sea, c1520 to the present. Helgoland Marine Research 59:205.
ICES (2001) Report of the Working Group on the Assessment of Demersal Fish Stocks in the North Sea and Skagerrak. ICES CM 2001/ACFM: 07
ICES (2015) Report of the Working Group on Crangon Fisheries and Life History (WGCRAN). 18-20 May 2015, Ijmuiden, the Netherlands. ICES CM 2015/SSGEPD: 07.58 pp.
ICES (2012) Report of the Working Group on the Assessment of Demersal Stocks in the North Sea and Skagerrak (WGNSSK). ICES Document CM 2012/ACOM: 13.
ICES (2012). Report of the Working Group on Crangon Fisheries and Life History (WGCRAN).
ICES (2013). Workshop on the necessity of a crangon and cephalopod management (WKCCM). ICES CM 2013/ACOM:82.
Jensen K.T. & Jensen J.N. (1985) The importance of some epibenthic predators on the density of juvenile benthic macrofauna in the Danish Wadden Sea. Journal of Experimental Marine Biology and Ecology 89: 157-174.
Kamermans P.K., C. Smit, Wijsman J. & Smaal A.C. (2013) Meerjarioge effect- en productiemaetingen aan MZI’s in de Waddenzee, de Oosterschelde en de Voordelta, samenvattend eindrapport. Imares C191/13.
Kuipers B.R. & Dapper R. (1984) Nursery function of Wadden Sea tidal flats for the brown shrimp Crangon crangon. Marine Ecology Progress Series 17: 171-181.
Leonard G.H., Bertness M. D. & Yund P. O. (1999) Crab predation, waterborne cues, and inducible defences in the blue mussel, Mytilus edulis. Ecology 80(1): 1-14.
Marsh, C.P. (1986) Rocky intertidal community organization: the impact of avian predators on mussel recruitment. Ecology 67(3): 771-786.
Mollet F.M., Dieckmann U. & Rijnsdorp A.D. (2016) Reconstructing the effects of fishing on life-history evolution in North Sea plaice Pleuronectes platessa. Marine Ecology Progress Series 542: 195-208.
Myers R., Hutchins J.A. & Barrowman N.C. (1996) Hypotheses for the decline of cod in the North Atlantic. Marine Ecology Progress Series 138: 293-308.
Nehls G., Hertzler I. & Scheiffarth G. (1997) Stable mussel Mytulis edulis beds in the Wadden Sea. They’re just for the birds. Helgoländer Meeresuntersuchungen 51(3); 361-372.
Nehls G. & Ruth M. (2004) Miesmuschelmonitoring und Miesmuschelmanagement im Nationalpark ‘‘Schleswig-Holsteinisches Wattenmeer’’Berichtszeitraum 1997–2002. Bericht an das Landesamt für den Nationalpark Schleswig-Holsteinisches Wattenmeer und das Amt für Ländliche Raume, Kiel.
Nehls G., Witte S., Dankers N., de Vlas J., Quirijns F. & Kristensen P. S. (2009) Fisheries. Thematic Report No. 3.3. In: H. Marencic & J. de Vlas (Eds). Quality Status Report 2009. Wadden Sea Ecosystems No 25.
Nilsson P., Sundback K. & Jonsson B. (1993) Effect of the brown shrimp Crangon crangon L. on endobenthic macrofauna, meiofauna and meiofaunal grazing rates. Netherlands Journal of Sea Research 31:95-106.
O'Neill F.G. & Ivanović A. (2016) The physical impact of towed demersal fishing gears on soft sediments. Journal of Marine Science 73: 5-14.
Pihl L. & Rosenberg R. (1984) Food selection and consumption of the shrimp Crangon crangon in some shallow marine areas in western Sweden. Marine Ecology Progress Series 15: 159-168.
Polet H., Depestele J. & van Craenest K. (2010) LOT 3, Scientific advice concerning the impact of the gears used to catch plaice and sole, Summary Studies and pilot projects for carrying out the common fisheries policy Ostend. Instituut voor Landbouw- en Visserijonderzoek 28: 23.
Pope J.G. & Macer C.T. (1996) An evaluation of the stock structure of North Sea cod, haddock and whiting since 1920, together with consideration of the impacts of fisheries and predation effects on their biomass and recruitment. ICES Journal of Marine Science 53, 1157–1169.
Revill A., Pascoe S., Radcliffe C., Riemann S., Redant F., Polet H., Damm U., Neudecker T., Kristensen P. S. & Jensen D. (1999) Economic consequences of discarding in the crangon fisheries. Final Report to the European Commission, 118.
Rijnsdorp A. D. (1989) Maturation of male and female North Sea plaice (Pleuronectes platessa L.). Journal du Conseil International pour l’Exploration de la Mer, 46: 35–51.
Schultz S., Günther C., Santos J., Berkenhagen J., Bethke E., Hufnagl M., Kraus G., Limmer B., Stepputtis D., Temming A. & Neudecker T. (2015) Optimierte Netz-Steerte für eine ökologisch und ökonomisch nachhaltige Garnelenfischerei in der Nordsee (CRANNET). Projektabschlussbericht. Hamburg; Rostock: Johann Heinrich von Thünen-Institut; Universität Hamburg, Institut für Hydrobiologie und Fischereiwissenschaften, 374 p.
Siegel V., Gröger J., Neudecker T., Damm U. & Jansen S. (2005) Long-term variation in the abundance of the brown shrimp Crangon crangon (L.) population of the German Bight and possible causes for its interannual variability. Fisheries Oceanography 14: 1-16.
Smaal A.C., Craeymeersch J., Drent J., Jansen J.M. & van Stralen M.R. (2013) Effecten van mosselzaadvisserij op sublitorale natuurwaarden in de westelijke Waddenzee: Samenvattend eindrapport. Imares C006/13.
Soetaert M., Decostere A., Polet H., Verschueren B. & Chiers K. (2015) Electrotrawling: a promising alternative fishing technique warranting further exploration. Fish and Fisheries 16: 104-124.
Temming A., & Hufnagl M. (2014) Decreasing predation levels and increasing landings challenge the paradigm of non-management of North Sea brown shrimp (Crangon crangon). ICES Journal of Marine Science, doi: 10.1093/icesjms/fsu194.
Tiews K. (1954) Die biologischen Grundlagen der Büsumer Garnelenfischerei. Berichte der deutschen wissenschaftlichen Kommission für Meeresforschung 13: 235-269.
Tulp I., Bolle L.J., Meesters E. & de Vries P. (2012) Brown shrimp abundance in northwest European coastal waters from 1970 to 2010 and potential causes for contrasting trends. Marine Ecology Progress Series 458: 141-154.
Tulp I., Chun C., Haslop H., Schulte K., Siegel V., Steenbergen J., Temming A. & Hufnagl M. (2016) Annual brown shrimp (Crangon crangon) biomass production in Northwestern Europe contrasted to annual landings. ICES Journal of Marine Science doi:10.1093/icesjms/fsw141.
van Beek F. A. (1990) Discards sampling programme for the North Sea Dutch participation. Internal Report Netherlands Institute for Fisheries Research, DEMVIS 90–303. 24 pp.
van der Heide T., Tielens E., van der Zee E.M., Weerman E.J., Holthuijsen S., Eriksson B.K., Piersma T., van de Koppel J., Olff H. (2014). Predation and habitat modification synergistically interact to control bivalve recruitment on intertidal mudflats. Biological Conservation 172: 163-169.
van Stralen M.R. (2016) Invang van mosselzaad in MZI’s. Resultaten 2015. Buro MarninX.
Verschueren B., Vanelslander B. & Polet H. (2012) Verduurzaming van de garnalenvisserij met de garnalenpuls: eindrapport. ILVO mededelingen, ILVO, Institute for Agricultural and Fisheries Research, 102 pp..
Walter, U. & de Leeuw D. (2007) Miesmuschel-Langleinenkulturen – Vom wissenschaftlichen Experiment zur wirtschaftlichen Umsetzung. Informationen aus der Fischereiforschung. 54:34-39.
WWF (2014) Sustainable brown shrimp fishery- is pulse fishing an option? WWF Deutschland, Berlin. https://www.wwf.de/fileadmin/fm-wwf/Publikationen-PDF/Technical-Report-….
WWF (2016) Wo die Krabben gefischt werden- Räumliche Verteilung und zeitliche Entwicklung bei der Nutzung des Wattenmeeres und der angrenzenden Nordsee durch die deutsche Krabbenfischerei von 2007 – 2013. WWF Deutschland, Berlin.
This report should be cited as: Baer, J., Smaal, A., van der Reijden, K., & Nehls, G. (2017). Wadden Sea Quality Status Report: Fisheries. Common Wadden Sea Secretariat. https://doi.org/10.5281/zenodo.15225400
All 2017 reports may be cited collectively as: Kloepper, S., Baptist, M. J., Bostelmann, A., Busch, J.A., Buschbaum, C., Gutow, L., Janssen, G., Jensen, K., Jørgensen, H.P., de Jong, F., Lüerßen, G., Schwarzer, K., Strempel, R., & Thieltges, D. (2017). Wadden Sea Quality Status Report. Common Wadden Sea Secretariat, Wilhelmshaven, Germany. Downloaded DD.MM.YYYY. qsr.waddensea-worldheritage.org